Melonite Processing
What is Melonite® Nitrocarburizing?
Melonite® is a thermochemical treatment for improving the surface properties of steel parts. It exhibits predictable and repeatable results in the treating of low and medium carbon steels, alloy steels, stainless and austenitic steels, tool and die steels, cast and sintered iron.
As the first job shop on the West Coast to offer Melonite processing, Burlington Engineering, Inc. uses its salt bath nitriding experience to diversify its servicing to the Southern California metal finishing industry. The system has many stages, from the pre-treatment-cleaning, to pre-heat furnace, to the Melonite salts, oxidizing salts, and water-cooled and rinsed.
Both Melonite and Melonite QPQ are used to improve the surface properties of plain carbon, low alloy, and stainless steels. These processes are categorized as Controlled Liquid Ionic Nitriding (CLIN), Salt Bath Nitriding (SBN), Ferritic Nitro-Carburizing (FNC) and include:
Melonite Processing
- Improved Corrosion Resistance
- Abrasive Wear Resistance
- Improved Scuffing Wear
- Increased Fatigue Strengths
- Black Color
How Does the Melonite® Nitriding Process Work?
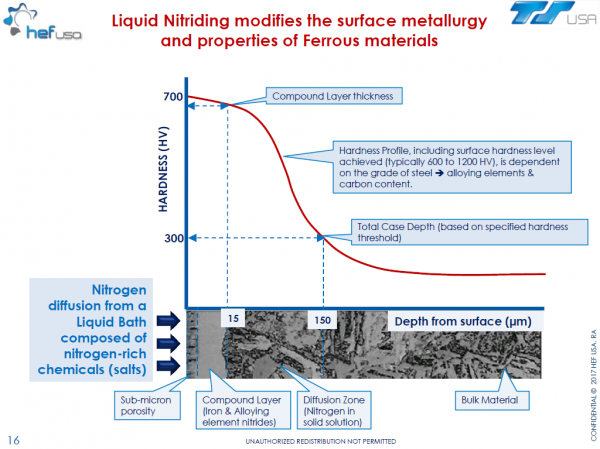
During the salt bath nitriding process, the metal surface is enriched with nitrogen and carbon. A two-part nitride layer consisting of a monophase compound layer and a diffusion layer is formed. Total depth ranges from 0.008-0.040" depending on the composition of the base material and treating time. Hardness in the compound layer ranges from approximately HV 700 on alloyed steels to HV 1600 on high chromium steels.
Zone 1: Compound Layer
The nitrogen-rich inter-metallic compound layer mainly contains iron-carbonitrides and, depending on the type and proportion of alloying elements in the base material, special nitrides.
A unique feature of salt bath nitrocarburized layers is the monophase ε-FexN compound layer, with a nitrogen content of 6-9% and a carbon content of around 1%. Compared with double phase nitride layers which have lower nitrogen concentrations, the monophase ε-FexN layer is more ductile and gives better wear and corrosion resistance by improvement with case hardening.
In the metallographic analysis the compound layer is clearly definable from the diffusion layer as a lightly etched layer. A porous area develops in the outer zone of the compound layer. The case hardness of the compound layer measured on a cross-section is around 700 HV for unalloyed steels and up to about 1600 HV on high chromium steels. Treatment durations of 1-2 hours usually yield compound layers about 10-20 µm thick (0.0004 - 0.0008"). The higher the alloy content, the thinner the layer for the same treatment cycle.
Zone 2: Diffusion Layer
The diffusion zone demonstrates a decreasing gradient concentration of carbon and particularly nitrogen as the gradient extends deeper into the surface of the substrate. This property yields a tough outer surface or shell, yet alloys the material to retain ductility, thereby lending to the overall strength of the material.
In unalloyed steels, the nitrogen is dissolved in the iron lattice. Due to the diminishing solubility of nitrogen in iron during slow cooling, γ'-Fe4N nitrides are precipitated in the outer region of the diffusion layer, some in the form of needles, which are visible in the structure under the microscope. If cooling is done quickly, the nitrogen remains in a super-saturated solution. With alloyed steels that contain nitride-forming elements, the formation of stable nitrides or carbonitrides takes place in the diffusion layer independent of the cooling speed.
With increasing alloy content of the steel, the diffusion layer is thinner for identical nitrocarburizing parameters. However, with their higher level of nitride-forming alloying elements, these steels have a greater case hardness.
Liquid Nitriding Modifies the Surface Metallurgy and Properties of Ferrous Metals
Contact Us to Learn More About Using Melonite® Salt Bath Nitriding
Burlington Engineering, Inc. provides salt bath nitriding, and other metal processing services for customers throughout California and beyond. We work with a wide range of industries and have the expertise to help you select the best process for your metal finishing project. Contact us today!

